|
In this situation it is generally uncommon to talk about half-life in the first place, but sometimes people will describe the decay in terms of its "first half-life", "second half-life", etc., where the first half-life is defined as the time required for decay from the initial value to 50%, the second half-life is from 50% to 25%, and so on. In a decay process that is not even close to exponential, the half-life will change dramatically while the decay is happening. The term "half-life" is almost exclusively used for decay processes that are exponential (such as radioactive decay or the other examples above), or approximately exponential (such as biological half-life discussed below). 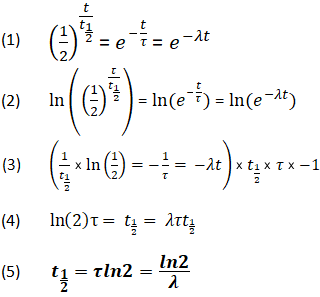
In a first-order reaction the half-life of the reactant is ln(2)/ λ, where λ (also denoted as k) is the reaction rate constant. In a chemical reaction, the half-life of a species is the time it takes for the concentration of that substance to fall to half of its initial value. 
For this example the term half time tends to be used rather than "half-life", but they mean the same thing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

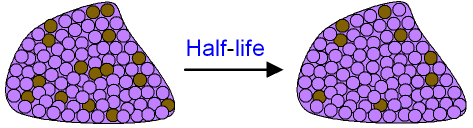
 RSS Feed
RSS Feed
